Mesenchymal stem cells (MSCs) are an appealing cell type for use in therapies for ischemia, but clinical trials have not yielded consistent benefits for patients. In their current form, MSC therapies are ineffective and unreliable, particularly for patients who have suffered a myocardial infarction. Work done by Dr. Baker and his team overcomes many of current limitations to MSC therapeutic approaches, including reduction of therapeutic activity by aging and disease, patient-to-patient variability in response, and MSC heterogeneity.
Through patient-specific enhancement of biomechanical pharmacological conditions, Dr. Baker has improved MSC therapy for the treatment of wound healing, peripheral ischemia, myocardial infarction and vascular grafts. His group has developed a platform to apply mechanical strain and small molecule inhibitors to induce MSC pluripotency without the need for transgenes, in an efficient, high throughput system (HT-BOSS1.) These optimally conditioned MSCs exhibit enhanced pericyte and endothelial behavior and show increased therapeutic potential in enhancing blood vessel growth in animal models of subcutaneous implantation and hind limb ischemia.
The University of Texas at Austin is seeking industry partnership to further develop the technology for therapeutic applications. US patent applications have been filed.
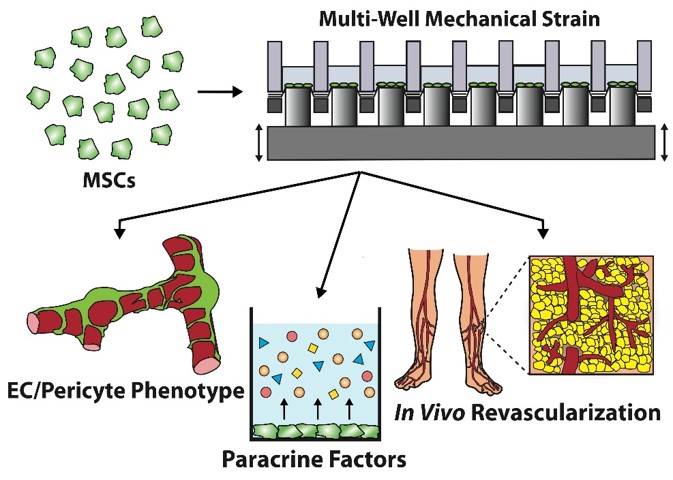
Following a combination of mechanical and pharmacological conditioning, MSCs begin to express markers for both pericytes and endothelial cells. These conditioned MSCs also exhibit superior properties in enhancing endothelial tube formation, production of paracrine or angiogenic growth factors, and induction of angiogenesis following implantation.

