This technology uses highly engineered T-cell receptors to precisely detect and target cytomegalovirus-infected cells, offering improved therapies and diagnostics by enhancing binding strength, stability, and resistance to viral immune evasion.
Background
Cytomegalovirus (CMV) is a widespread herpesvirus that poses significant health risks, particularly to immunocompromised individuals such as transplant recipients, cancer patients, and those with HIV/AIDS. CMV infection can lead to severe complications, including organ rejection, encephalitis, and even death. The immune system’s natural response to CMV relies heavily on T cells recognizing viral peptides presented by major histocompatibility complex (MHC) molecules, especially the immunodominant pp65 peptide (NLV) presented by HLA-A2*02:01. However, in many patients, the endogenous T-cell response is insufficient to control viral replication, necessitating the development of targeted immunotherapies and sensitive diagnostic tools to detect and manage CMV infections effectively.
Current approaches to targeting CMV-infected cells using natural T-cell receptors (TCRs) are limited by several factors. Wild-type TCRs typically exhibit low affinity for their peptide-MHC targets, which restricts their therapeutic efficacy and sensitivity as diagnostic reagents. Furthermore, when expressed as soluble proteins, natural TCRs often suffer from poor stability and low yields, complicating their use in clinical and laboratory settings. Compounding these challenges, CMV encodes viral Fc receptors that can bind and sequester therapeutic antibodies or Fc fusion proteins, thereby undermining immune clearance and reducing the effectiveness of antibody-based therapies. These limitations highlight the need for improved molecular tools that combine high specificity, enhanced affinity, and resistance to viral immune evasion mechanisms.
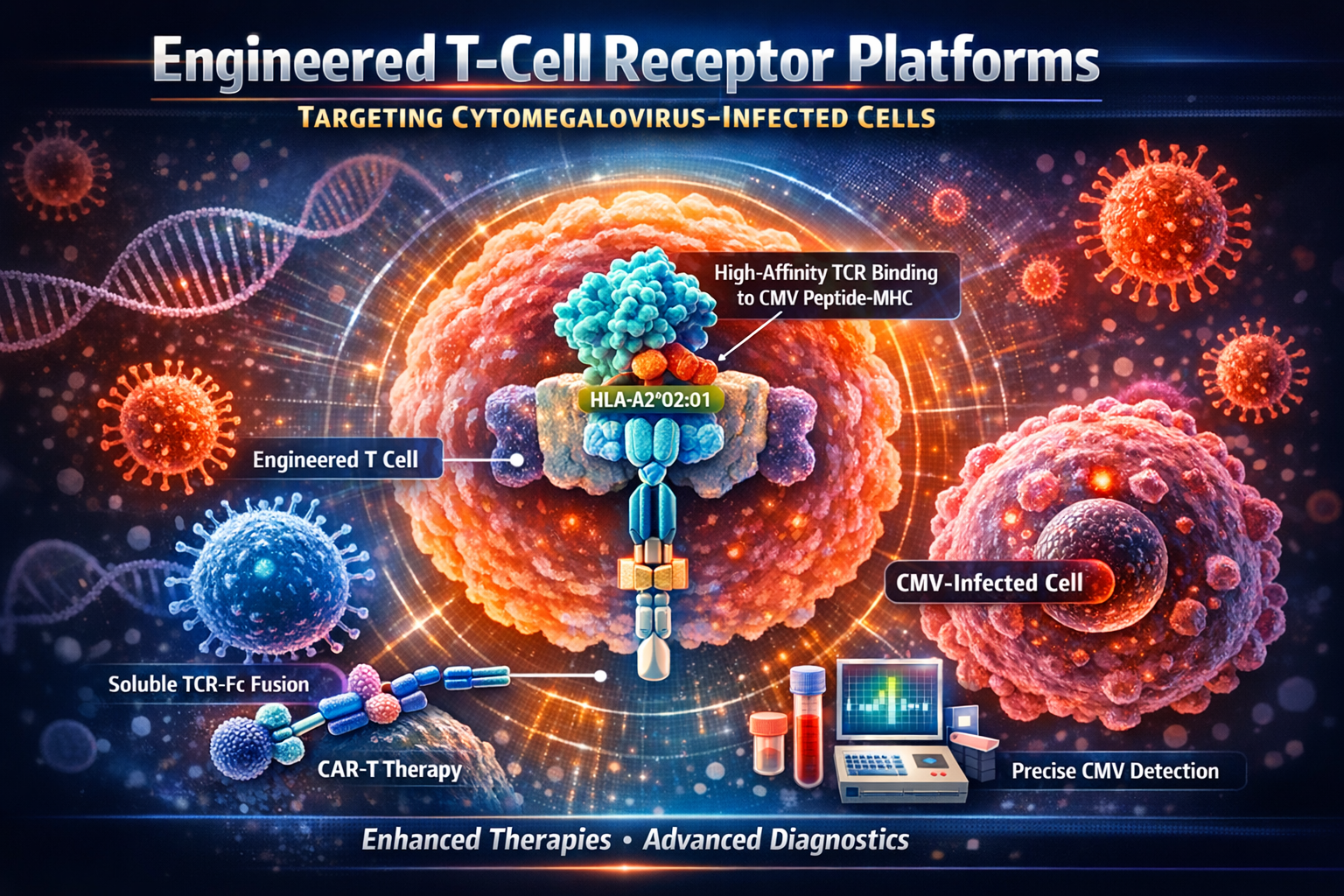
Technology Description
This technology centers on the development of high-affinity engineered T-cell receptors (TCRs) specifically designed to recognize the cytomegalovirus (CMV) pp65 peptide (NLVPMVATV) presented by the HLA-A2*02:01 molecule. Utilizing structure-guided mutagenesis and mammalian cell surface display, the TCRs are optimized for nanomolar to sub-nanomolar affinity, far surpassing the binding strength of natural TCRs. These receptors can be configured as membrane-bound, soluble TCR-Fc fusion proteins, or incorporated into chimeric antigen receptors (CARs) and bispecific molecules. Soluble TCRs are further engineered for stability and homogeneity through the introduction of disulfide bonds and removal of glycosylation sites, while the Fc domain is modified to reduce binding to CMV-encoded viral Fc receptors without compromising interaction with host immune receptors. The technology supports a wide range of applications, including adoptive cell therapies, sensitive diagnostic assays, and disease monitoring.
What differentiates this technology is its comprehensive engineering strategy that addresses both affinity and functional resilience in the context of viral immune evasion. By leveraging mammalian cell display and targeted CDR3 loop mutagenesis, the TCRs achieve exceptional specificity and stability, validated through rigorous biophysical assays. The unique Fc engineering ensures that therapeutic and diagnostic agents remain effective even in the presence of CMV’s viral Fc receptors, a common mechanism of immune escape. Additionally, the modular design allows integration into various therapeutic formats—ranging from engineered immune cells to bispecific engagers—enabling tailored interventions for CMV infections and CMV-associated malignancies. This versatility, combined with the ability to detect low-abundance antigen presentation and compatibility with combination immunotherapy protocols, establishes the platform as a robust and differentiated solution for tackling CMV-related diseases.
Benefits
- Significantly enhanced affinity and specificity for CMV pp65 peptide presented by HLA-A2*02:01, enabling precise targeting of CMV-infected cells.
- Improved stability and homogeneity of soluble TCR-Fc fusion proteins through engineered disulfide bonds and glycosylation site removal.
- Engineered Fc domains reduce binding to viral Fc receptors, overcoming CMV immune evasion while maintaining host immune effector functions.
- Versatile formats including membrane-bound TCRs, soluble TCR-Fc fusions, CARs, and bispecific molecules for diverse therapeutic and diagnostic applications.
- Enables adoptive cell therapies using engineered T cells, NK cells, or stem cells for treatment of CMV infections and CMV-associated cancers.
- High sensitivity detection of CMV peptide-MHC complexes for monitoring disease progression and vaccine efficacy.
- Compatibility with combination therapies such as lymphodepletion, cytokines, and immune checkpoint inhibitors to enhance clinical outcomes.
Commercial Applications
- CMV adoptive T cell therapy
- CMV diagnostic assays
- CMV-targeted bispecific T cell engagers
- CMV-specific CAR-T cell development
- Monitoring CMV vaccine efficacy
Opportunity
This patented technology is available for exclusive licensing to a commercial partner. High-affinity engineered T-cell receptors (TCRs) target the CMV pp65 peptide presented by HLA-A2*02:01. Developed via structure-guided mutagenesis and mammalian cell display, these TCRs achieve nanomolar binding. They can be membrane-bound, soluble (TCR-Fc fusions with stabilized, deglycosylated, and viral FcR-evading Fc domains), or CARs. Applications include disease treatment, monitoring, and cellular detection of specific peptide presentation.
Patent
High Affinity Engineered T-Cell Receptors Targeting Cmv Infected Cells
US2022144916A1
Publication
Weaponizing T-cell receptors through molecular engineering

