Drug-Eluting Porous Polymer Tubes for Sustained Local Therapeutic Delivery in Medical Devices
A porous polyethylene chest tube releases pain medication locally for up to two weeks after thoracic surgery, enhancing pain management while maintaining the flexibility and functionality of standard tubes.
Background
Effective pain management is crucial for patients undergoing thoracic surgery, as inadequate control can lead to complications such as impaired breathing, prolonged hospital stays, and reduced overall recovery. Traditional methods often rely on systemic analgesics, which may not provide sufficient localized pain relief and can cause unwanted side effects like sedation, nausea, and respiratory depression. Additionally, the use of standard chest tubes does not address the need for sustained, targeted delivery of analgesics directly to the surgical site, limiting the effectiveness of pain control and patient comfort during the postoperative period.
Current approaches to managing post-thoracic surgery pain with standard chest tubes present several challenges. These tubes typically lack the capability to deliver drugs in a controlled and sustained manner, resulting in fluctuating pain relief and the necessity for frequent dosing. Systemic administration of analgesics can lead to inconsistent pain management and increase the risk of adverse effects. Moreover, existing chest tubes may compromise patient mobility and comfort due to their rigidity and lack of flexibility. The inability to maintain mechanical integrity while providing effective pain control highlights the need for improved chest tube designs that integrate localized drug delivery systems, ensuring both enhanced patient outcomes and streamlined postoperative care.
Technology Description
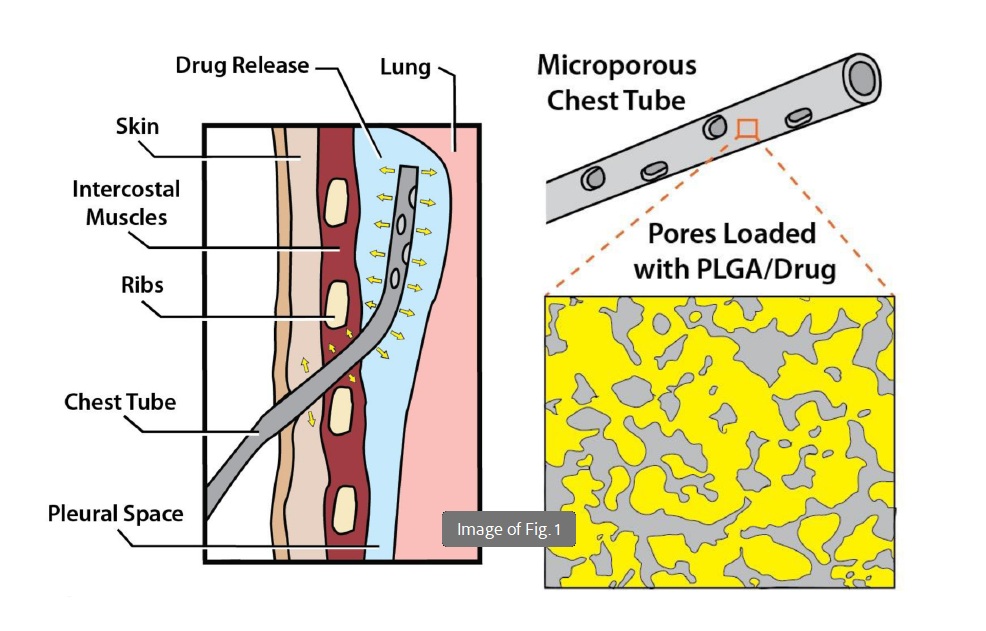
This drug-eluting chest tube utilizes sintered ultra-high molecular weight polyethylene (UHMWPE) available in three variants—ST1, ST2, and ST3—with differing porosity and pore sizes. The ST3 variant features the highest porosity and lowest density, enhancing its compliance compared to standard chest tubes. It is engineered to deliver bupivacaine locally through two primary methods: embedding microparticles within its pores or employing a solid-in-solid casting technique with PLGA and bupivacaine. The solid-in-solid approach, particularly in the ST3 variant, achieves a more sustained and higher drug release. Comprehensive characterization using scanning electron microscopy and microCT confirms the interconnected pore structure, while mechanical testing demonstrates that ST3 maintains flexibility akin to commercial PVC tubes, even when loaded with PLGA. This design supports controlled, therapeutically relevant anesthetic delivery for up to 14 days, making it highly effective for post-thoracic surgery pain management.
What sets this technology apart is its superior drug delivery mechanism and material optimization. The solid-in-solid loading method not only ensures a consistent and controlled release of bupivacaine but also allows for higher drug loading capacity compared to traditional microparticle methods. The ST3 variant’s unique combination of high porosity and flexibility ensures that the mechanical integrity of the chest tube is preserved, enhancing patient comfort and device functionality. Additionally, the ability to reload the drug and the versatility in delivering multiple therapeutic agents extend its applicability across various medical devices, including endotracheal tubes and surgical drains. This advanced system offers significant improvements in patient outcomes by providing sustained, localized drug delivery while maintaining essential device performance.
Benefits
- Provides controlled and sustained release of bupivacaine for up to 14 days
- Maintains mechanical flexibility comparable to standard PVC chest tubes
- Offers higher drug loading capacity and more efficient drug release using the solid-in-solid approach
- Protects the drug payload during insertion, ensuring consistent therapeutic delivery
- Enhances patient comfort with flexible regions and variable stiffness sections
- Allows for potential in situ drug reloading for extended therapy
- Broad applicability across various medical devices, including endotracheal tubes and Foley catheters
- Supports delivery of multiple therapeutic agents, such as antibiotics and anti-viral agents
- Ensures interconnected pore structures for optimal drug distribution and release kinetics
- Facilitates manufacturing scalability and material optimization for diverse clinical applications
Commercial Applications
- Post-surgical chest drainage
- Surgical drain pain management
- Endotracheal tube analgesia
- Foley catheter drug delivery
- Orthopedic implant analgesia
Opportunity
This patent is available for exclusive licensing. A chest tube made from sintered ultra-high molecular weight polyethylene in three variants, with ST3 offering highest porosity and flexibility. It delivers bupivacaine locally using a solid-in-solid PLGA embedding method, enabling controlled release for up to 14 days. Characterized by interconnected pores via SEM and microCT, it maintains mechanical properties comparable to PVC tubes. Additional features include dual-tube designs and variable stiffness sections, suitable for various medical devices requiring sustained therapeutic agent delivery.
Patent
US20240245834A1

